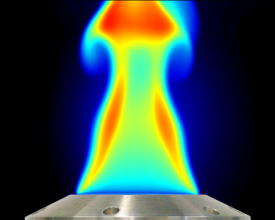
Emission Spectroscopy: Spectral Imaging
Based on emission spectroscopy flame emission is analyzed using spectral imaging. Flame emission imaging is a line-of-sight method like absorption and probes electronically excited species in contrast to LIF which probes ground state species. 2D flame species imaging is carried out with a set of filters mounted in front of the camera lens or 1D using an imaging spectrograph for correlated multi-species detection.
The flame emission (chemiluminescence) of flame radicals like OH*, CH*, C2* is used to e.g. visualize the reaction zone or to monitor flame stoichiometry via emission ratios of such radicals. Infrared emission visualizes major flame components like unburnt fuel, CO2 or water.
2-color pyrometry (blackbody radiation) is applied to measure temperature fields in sooty flames. Temporal flame analysis (flame flicker) is measured using statistical information from a time series of flame images. High speed imaging is applied for flame propagation and extinction measurements.
Absorption Spectroscopy
Absorption Spectroscopy is one of the oldest techniques for the non-intrusive investigation of gaseous media. This technique is based on the absorption of radiation emitted by a spectrally well defined light source along its way to the detector. The loss of light intensity is a measure for the absorbing gas concentration using the Lambert-Beer law. The more gas molecules in the light path, the more light will be absorbed. LaVision's absorption based sensors measure precisely the drop of light intensity and calculates the molecule's concentration from this value. The absorption wavelength is specific for the probed molecule, i. e. the choice of the absorption wavelength determines on the type of detected molecule.
There are basically two different absorption techniques: broadband absorption and tunable laser absorption spectroscopy: Broadband absorption is used for the concentration measurement of functional groups of molecules like for the total hydrocarbon (fuel) concentration measurement inside an engine cylinder. The achievable sampling rates can be much higher compared to conventional gas analysis systems.
Tunable Diode Laser Absorption Spectroscopy (TDLAS) is highly sensitive and selective for the detection of smaller molecules such as H2O, O2 or CO2. This technique measures the concentration of the molecules, but also the temperature and/or the total ambient pressure of the bath gas using sophisticated spectral analysis.

